 
The former definition of the mole was that a mole was 12 g of 12C contains 1 mole of 12C atoms (its molar mass is 12 g/mol). Per the amu definition, a single 12C atom weighs 12 amu (its atomic mass is 12 amu). (credit: modification of work by Mark Ott).īecause the definitions of both the mole and the atomic mass unit are based on the same reference substance, 12C, the molar mass of any substance is numerically equivalent to its atomic or formula weight in amu. From left to right (bottom row): 32.1 g sulfur, 28.1 g silicon, 207 g lead, and 118.7 g tin. From left to right (top row): 65.4 g zinc, 12.0 g carbon, 24.3 g magnesium, and 63.5 g copper. CO 2 has one carbon atom and two oxygen atoms.The most common of these being salt, or sodium chloride, and the potassium compounds sylvite (or potassium chloride) and carnallite (potassium magnesium chloride hexahydrate). Oxygen (O) has an atomic mass of about 16.00 amu. Chlorine is in group 17 of periodic table, also called the halogens, and is not found as the element in nature - only as a compound.With aluminium, antimony and copper it forms highly magnetic alloys. Drinks cans are made of an alloy of aluminium with 1.5 manganese, to improve resistance to corrosion. This is extremely strong and is used for railway tracks, safes, rifle barrels and prison bars. Carbon (C) has an atomic mass of about 12.01 amu. Manganese steel contains about 13 manganese.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound.Molar mass of carbon: 12.01 g/mol Molar mass of hydrogen: 1.008 g/mol Step 4: For each element, multiply its molar mass by the. Multiply the subscript (number of atoms) times the atomic mass of that element and add the masses of all of the elements in the molecule to get the molecular mass. Step 3: Find the molar mass of each element from the periodic table. 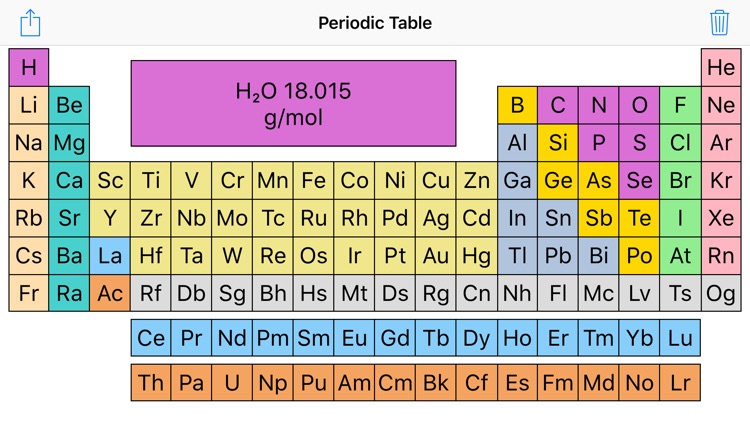
The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Find the atomic mass for each element by using the mass given in the Periodic Table or table of atomic weights. Find atomic masses: look up the atomic masses of each element present in the compound.For example, water is H 2O, meaning it contains two hydrogen atoms and one oxygen atom. Identify the compound: write down the chemical formula of the compound.One mole contains exactly 6.022 ×10 23 particles (Avogadro's number) Mole is a standard scientific unit for measuring large quantities of very small entities such as atoms and molecules.Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol.(1 u is equal to 1/12 the mass of one atom of carbon-12) Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).For example, if we are trying calculate for ammonia (NH 3 ), then we. The atomic mass is equal to the atomic number which is listed below the element symbol. To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations: Molar Mass of Ammonia NH 3 Step 1: The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Tos, Bz, TMS, tBu, Bzl, Bn, DmgĮxamples of molar mass computations: NaCl, Ca(OH)2, K4, CuSO4*5H2O, nitric acid, potassium permanganate, ethanol, fructose, caffeine, water.Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Enter a chemical formula to calculate its molar mass and elemental composition:Ĭomputing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
